This article provides an understandable and concise explanation about the structure and function of nucleic acids, their monomers and polymers.
Key takeaways:
- Nucleic acid monomers consist of nitrogenous bases, sugar molecules, and phosphate groups.
- Nucleotides are the building blocks of genetic material.
- Nucleotide components include nitrogenous bases, sugars, and phosphate groups.
- Nucleic acid polymers DNA and RNA have different functions and structures.
- Polymers exhibit enhanced properties compared to their monomers.
What You Will Learn
Nucleic Acid Monomers
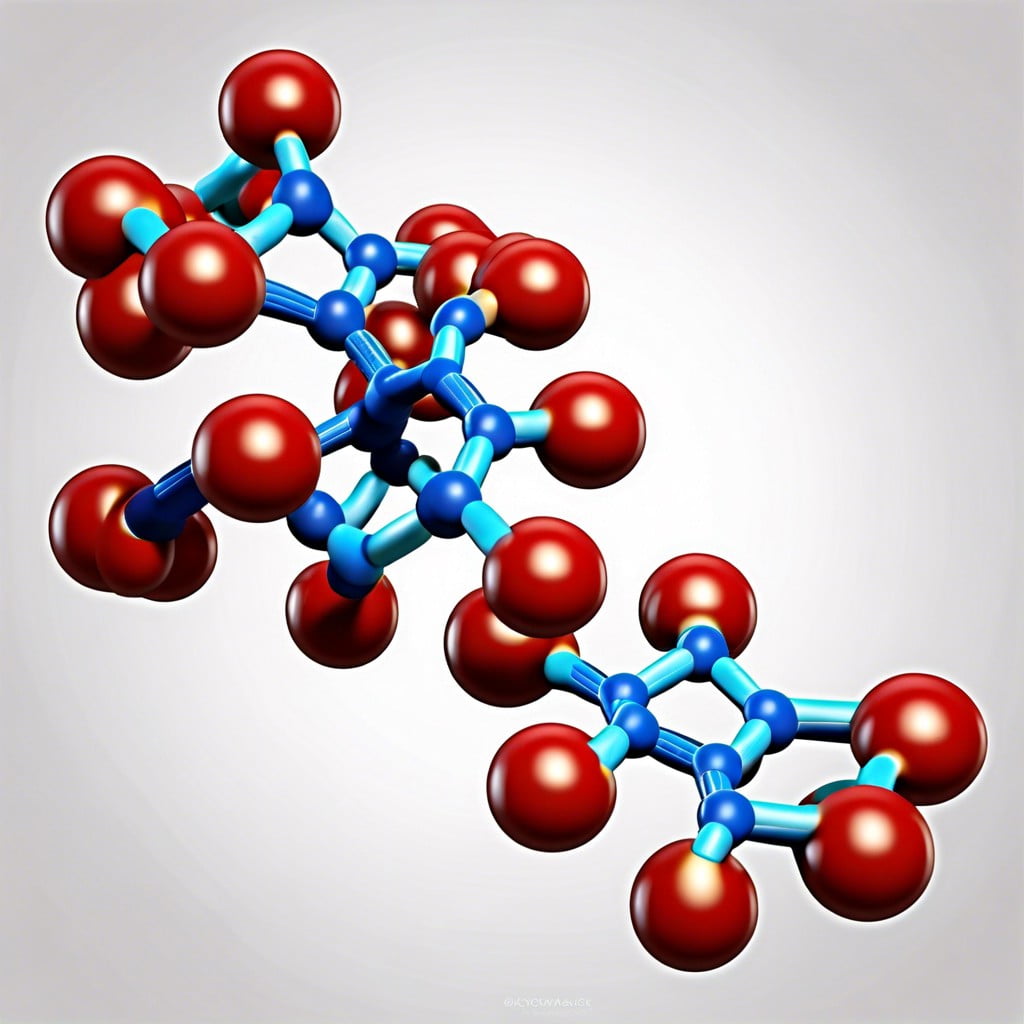
Understanding the building blocks of nucleic acids begins with a close look at nucleotides. Each nucleotide consists of three essential components: a nitrogenous base, a sugar molecule, and a phosphate group.
The nitrogenous bases are molecules containing nitrogen, and they come in two types: purines (adenine and guanine) and pyrimidines (cytosine, thymine, and uracil). The sugar component is either ribose in RNA or deoxyribose in DNA, differing by just one oxygen atom. Lastly, the phosphate group attaches to the sugar, playing a pivotal role in connecting nucleotides together.
Once these pieces come together, they form the monomers ready to join into long, intricate chains, the backbone of genetic material.
Definition of Nucleotides
Nucleotides are the building blocks of life’s genetic material—they’re like the alphabet of genetics, each one an individual letter that joins to form the instructions within our cells.
Each nucleotide comprises three parts: a nitrogenous base providing its unique identity (adenine, guanine, cytosine, or thymine in DNA; add uracil for RNA), a sugar molecule (deoxyribose in DNA and ribose in RNA) that acts like the spine of the structure, and a phosphate group that’s akin to a clasping hand, connecting one nucleotide to the next in a chain.
Understanding nucleotides is crucial because they’re not just letters; they’re the foundation upon which the genetic language is built, determining everything from hair color to how our bodies fight disease.
Components of a Nucleotide: Nitrogenous Bases, Sugars, Phosphate
Nucleotides are akin to the building blocks of nucleic acids, which include DNA and RNA. They consist of three essential parts:
1. Nitrogenous Bases: These are molecules containing nitrogen and come in two types: Purines (adenine and guanine) and Pyrimidines (cytosine, thymine, and uracil). These bases are responsible for the genetic coding of traits, as their sequence forms the genetic instructions.
2. Sugars: A five-carbon sugar is part of every nucleotide. In DNA, the sugar is deoxyribose, while in RNA, the sugar is ribose. The difference lies in the presence of an oxygen atom in the ribose sugar, which is absent in deoxyribose.
3. Phosphate Group: This component consists of one phosphorus atom bonded to four oxygen atoms. It is this group that links together nucleotides through phosphodiester bonds, forming the backbone of DNA or RNA. The phosphate also plays a critical role in the transfer of energy within the cell, particularly in the form of the molecule ATP.
Understanding these components is vital, as their arrangement determines the type and properties of the nucleic acid formed.
Nucleic Acid Polymers
DNA and RNA serve as the cell’s command center and messengers. DNA, with its famous double helix structure, is stable and stores genetic blueprints long-term. RNA, typically single-stranded, is the active translator that carries instructions from DNA to the cell’s protein-making machinery.
Imagine DNA as an archive. It holds the master copies of instructions that are crucial for the cell’s operations. RNA takes on several forms – messenger RNA (mRNA), ribosomal RNA (rRNA), and transfer RNA (tRNA). Together, they help in deciphering the DNA code and guiding the production of proteins, which are the workhorses of the cell.
These nucleic acid polymers are not static; they undergo replication and transcription. During replication, DNA duplicates itself before a cell divides, ensuring each new cell receives an exact copy of genetic instructions. Transcription is the process by which a segment of DNA is copied into RNA, marking the first step in expressing a gene into a functional protein.
DNA and RNA: Fundamental Differences
DNA and RNA serve as the cell’s master blueprints and workers, respectively. DNA, or deoxyribonucleic acid, is stable and resides in the cell’s nucleus, acting as long-term storage for genetic information. Its double-stranded helix consists of a deoxyribose sugar backbone and four types of nitrogenous bases: adenine, thymine, cytosine, and guanine.
On the flip side, RNA, ribonucleic acid, typically consists of a single strand. It uses ribose sugar and swaps out thymine for uracil as one of the bases. RNA is more transient in nature, converting the genetic codes into instructions for protein assembly.
These fundamental differences allow DNA to maintain integrity over generations, while RNA translates and executes the genetic instructions, adapting to real-time cellular needs.
FAQ
What is the monomer of deoxyribonucleic acid polymer?
The monomer of deoxyribonucleic acid (DNA) polymer is a nucleotide.
What is an example of a monomer and a polymer?
Glucose is an example of a monomer, and it can link up to form polymers such as glycogen, starch, and cellulose.
What are the monomers that make up polymers?
The monomers that make up polymers are primarily sugars, amino acids, fatty acids, and nucleotides.
What are the five monomer units of a nucleic acid?
The five monomer units of a nucleic acid are uracil, cytosine, thymine, adenine, and guanine.
How are the monomers of nucleic acids connected to form a polymer?
The monomers of nucleic acids, also known as nucleotides, connect to form a polymer through a process termed polymerization, during which phosphodiester bonds are formed between the phosphate group of one nucleotide and the hydroxyl group on the sugar of another.
How do the properties of a polymer differ from its individual monomers?
Polymers exhibit a range of properties, such as increased strength, flexibility, and resistance to environmental conditions, that are significantly different and often enhanced compared to their individual monomer units.
What forms can polymers take in the fields of construction and engineering?
Polymers in construction and engineering can take several forms including concrete additives, structural resins, coatings, seals, adhesives, and insulation.
Related reading:





